New discovery explains why binge drinking leads to alcohol dependence and suggests new treatments
Neuroscientist, Amy Lasek, at the Department of Psychiatry at the University of Illinois at Chicago, and colleagues, report that after binge drinking, neurons in brain circuits responsible for alcohol addiction become encased in a protein material, called a perineuronal net. The impenetrable coating cements neurons involved in alcohol addiction into a circuit that is extremely difficult to break. Current drugs for treating alcohol dependence work by modifying neurotransmitter signaling between neurons, but for many people these treatments cannot break the overwhelming compulsion to drink. Drugs that can break down the glue-like cement in perineuronal nets could offer a new approach to treatment.
Lasek’s unusual approach into addiction research stems from her background as a molecular and cell biologist working in the field of cancer research. The root of cancer is changes in specific genes. Small molecules designed to target these aberrant genes is the approach used in cancer therapy. Lasek’s background made her think of finding molecularly targeted therapies for psychiatric disorders.
Lasek and her colleagues began by studying fruit flies to search for gene variations that altered the fly’s behavior toward alcohol. She found several genes that had this effect, including an obscure one called ALK (anaplastic lymphoma kinase). Then she suppressed these genes in mice to see if the animal’s response to alcohol was altered. “I got hooked,” she says, “because to me the fact that you can manipulate a single gene in a single brain region and change behavior—like drinking or cocaine reward—was fascinating from a biological point of view!”
She and her colleagues examined the genome of families who had a history of alcohol dependence. They found that ALK—the gene they had identified in fruit flies, which altered the insects’ responses to alcohol—was also associated with people in families with a history of alcohol dependence. Lasek found several variations in the ALK gene (polymorphisms) that were strongly associated with differences in the immediate reaction individuals had to drinking alcohol, such as the subjective high or the amount of motor uncoordination experienced after having a drink. This correlation was a strong clue that ALK and alcohol abuse were somehow linked.
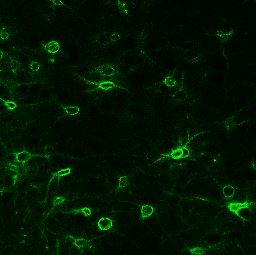
Surprisingly, it turned out that the protein made by the ALK gene was not controlling neurotransmitter signaling; it was on the surface of neurons where it controlled deposition of proteins called the extracellular matrix, which binds cells together into tissue. Some neurons are heavily encased in a special meshwork of extracellular matrix, called the perineuronal net. “It’s something like collagen,” she says. A very tough and slippery material that is resistant to change after it is deposited. But how could “brain glue” be related to alcoholism?
Research in other labs has shown that perineuronal nets are a specialized form of extracellular matrix that regulate synaptic plasticity—the ability of neurons to make and break connections between neurons. “You only get synapses [formed] where there are holes in the nets,” Lasek explains. When a neuron becomes heavily encased in a perineuronal net, new synapses cannot form and existing synapses become cemented in place.
Addiction is thought to be an aberrant learning process. Fundamentally, learning is connecting different events or environmental stimuli together to direct a specific behavior, such as Pavolv’s dogs learning to associate the sound of a bell with food to follow. At a cellular level, synaptic connections are formed and strengthened, weakened and broken, to encode the learning into a neural circuit that controls behavior. Likewise in addiction, a person learns to associate certain environmental stimuli or internal mental states, with an overpowering compulsion to drink alcohol. Quitting time, perhaps, may trigger an overpowering urge to have a cocktail, and then another, and another. “It may be that perineuronal nets are locking in that learning process that occurred, where you have this aberrant memory for the drug.”
Neurologist Varda Lev-Ram and colleagues, working at the University of California, San Diego, reported at the same meeting that perineuronal nets are extremely long lasting. She and her team discovered this by spiking mouse chow with traces of the isotope of nitrogen 15N, which would become incorporated into newly synthesized proteins in the animal’s body. Using this method of biological dating, the researchers found that some of the most long-lasting proteins in the body are components of the perineuronal nets.
These nets can be broken down and when this is done, long-term memory is eroded. When Lev-Ram and colleagues treated mice with compounds that inhibit enzymes that remodel components in the perineuronal nets (enzymes called matrix metalloproteinases), mice trained to fear a tone that signaled an electric shock, soon forgot the association between the warning tone and the electric shock. This is significant, because fear memories, as in PTSD, are some of the most difficult memories to break, but interfering with formation of the perineuronal net allows these traumatic memories to slip away.
Lasek’s team devised an experiment in which young adult mice were provided water tainted with alcohol in a manner similar to binge drinking in college-age students. When they examined brains of mice after six weeks of binge drinking they found that perineuronal nets formed and thickened around neurons in the insula, a part of the cerebral cortex that is known to be involved in compulsive alcohol use. These deposits did not develop around neurons in other regions of cerebral cortex, for example motor cortex, which controls bodily movement, suggesting a specific effect targeted on neurons involved in alcohol dependence. “These nets are accumulating in response to drugs of abuse. It may be locking in that learning process that occurred, where you have this aberrant memory for the drug,” she says.
This new finding suggests that to conquer addiction, “You have to get rid of the nets,” Lasek says. In her lab she is treating mice with inhibitors of ALK and other proteins in the perineuronal nets, and the unpublished results thus far show that these mice voluntarily decrease their binge drinking. “This would be an entirely new avenue of treatment,” she says. However, drug treatments are not the only way to take advantage of this new finding, because many other factors will influence how the perineuronal nets form and how quickly they can break down, including exercise and diet, she suggests. “I think these kind of things can ameliorate whatever risk you have with your genetics,” Lasek says. “I don’t always think that drugs are the solution; sometimes you need both [medicine and lifestyle changes].” Perineuronal nets are a new part of the puzzle explaining why it is so difficult to overcome alcohol addiction, and this new insight offers fresh hope for people whose lives are destroyed by addiction.
References
Chen, H., He, D., Lasek, A.W. (2015) Repeated binge drinking increases perineuronal nets in the insular cortex. Alcohol Clin. Exp. Res. October, 39:1930-8.
Chen, H., He, D., Lasek, A.W. (2015) Repeated binge drinking increases perineuronal nets in the insular cortex. Society for Neuroscience Meeting Abstract 695.01/N12
Lev-Ram, V., Bushong, E.A., Deerinck, T.J., Poczatek, C.J., Lechene, C.P., Palida, S.F., Taliman, K.M., Savas, J.N., Yates, J.R., Ellisman, M.H., Tsien, R.Y. (2015) Are very long-term memories stored in the pattern of holes in the perineuronal net? Society for Neuroscience Meeting Abstract 391.03/C27
Palida, S., Lev-Ram, V., Bushong, E.A., Ellisman, M.H., Tisen, R.Y. (2015) Visualizing structure and activity-dependent changes in the perineuronal net, a putative substrate for very long-term memory. Society for Neuroscience Meeting Abstract 391.04/C28
Modified from Scientific American: http://blogs.scientificamerican.com/guest-blog/why-binge-drinking-may-wire-the-brain-for-alcohol-dependence/