The Mystery Mark of Cancer
Brain cancer can be one of the most deadly forms of the disease, but all cancers are the result of uncontrolled cell division. Could all cancers share a common failure in the way genetic material is processed? Mysterious spots have been discovered in the nucleus of cells that marks them as being cancerous and they foretell the progression and severity of the disease. Scientists are tracking these puzzling cellular footprints found in all types of cancer to discover the mechanism of cancer and find a pathway to new treatments.
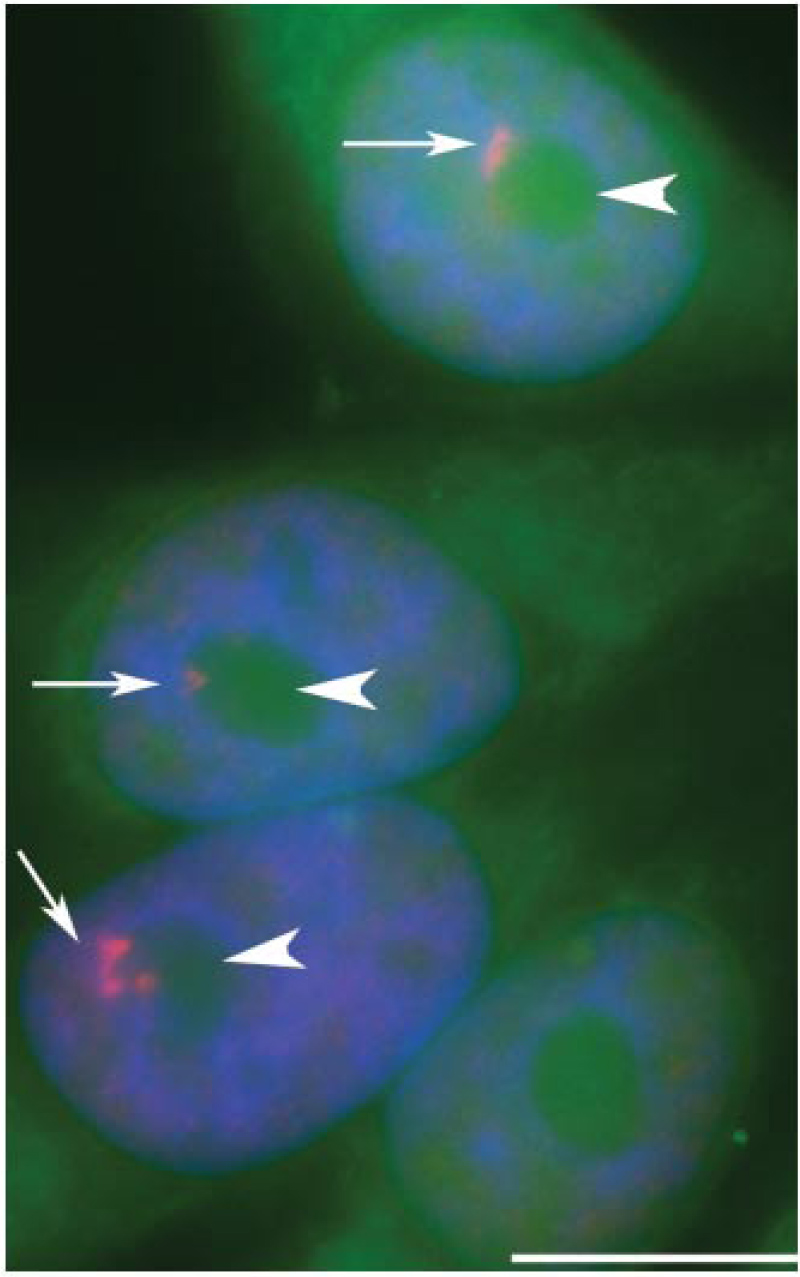
It looks as though the nucleus of cancer cells were dotted with a pink fluorescent felt pen. What these spots are is uncertain, but researchers have found the peculiar particles in all types of cancer cells, from breast to liver cancer. The spots, called perinucleolar compartments (PNC), were detected by Dr. Sui Huang of Northwestern University by injecting nuclei extracted from cancer cells into mice to generate antibodies. She then applied the antibodies to tissue samples examined under a microscope and found that they tagged strange-looking spots adhering to the nucleolus, a well-known spherical structure inside the nucleus. Normal cells never showed these PNC structures in their nucleus.
Presenting her latest research last week at the 49th Annual Meeting of the American Society for Cell Biology in San Diego, cell biologist Sui Huang, from Northwestern University, reported that these particles are composed of abnormal aggregates of nuclear proteins and genetic material that normally control the operation of genes.
In studying breast cancer she found that the number of cells in a tumor that were marked with PNCs correlated with the stage of cancer and its tendency to spread. For all kinds of cancers this correlation held—the more aggressive the cancer, the higher the percentage of cells in it that were marked with PNCs.
“It’s a genuine puzzle,” she told me after her talk. “We are very intrigued by this novel RNA-protein interaction and what they are doing in there. This kind of partnership (between the proteins and ribozymes) has not been shown before. That’s why it is so exciting.”
In addition to proteins, the aggregates contain types of RNA that have catalytic activity (ribozymes). Normally this catalytic activity processes the newly minted RNA transcripts manufactured from the DNA code in our genes. After the new string of RNA is read out from a gene, it is cut and spliced by ribozymes into a mature form that becomes the template for assembling a specific protein. But the proteins she and her colleagues identified in combination with these ribozymes in PNCs were not previously known to associate with one another.
Asked what the structure might do she admitted intriguingly that, “I think this structure does something that we actually don’t even know—but that doesn’t stop us from using it.”
Although the markers could be used for diagnosis to identify cancer cells and stage them, Dr. Huang sees an even bigger opportunity. Since tumors that have more cells with PNCs in them are more likely to develop into deadly spreading metastatic cancers, then, she reasoned, anything that could reduce the formation of PNCs should stop tumors from growing and spreading. She simply applied a large number of different compounds to cancer cells searching for any that could inhibit the formation of PNCs. The strategy paid off with the discovery of an entirely new compound that shrinks tumors in mice dramatically.
“Now we have compounds—we don’t actually know what they do, we just know that they can get rid of the tumor! So it works, we don’t care how.”
The next step will be to determine how these compounds work and this will begin to reveal what PNCs are doing. “Even though it’s (going to be) difficult, we are hooked,” she laughed. “That’s the problem.”
Photo Credit: J. Cellular Biochemistry Vol 107, p. 190, (2009)